Niemann-Pick Disease Type C
A rare, fatal and progressive genetic disorder characterized by a defect in the NPC1 protein causing cholesterol and lipids to accumulate in cells of major organs leading to cell and tissue dysfunction and death.

*Scope: United States + 79 other countries; *Commissioned Tessellon Inc – former Kantar Health experts with 25+ years of epidemiology and forecasting experience, (www.Tessellon.com); *Exhaustive literature search with a broad range of MESH terms.
Trappsol® Cyclo™
A Potential Treatment for the Systemic and Neurological Symptoms of NPC
Trappsol® Cyclo™ is a proprietary formulation of hydroxypropyl beta cyclodextrin and in multiple clinical studies has shown encouraging results to effectively manage the transportation of cholesterol. Taking the place of the defective NPC1 protein, Trappsol® Cyclo™, with its cyclic structure, captures cholesterol within the cell and transports it out of the cell.
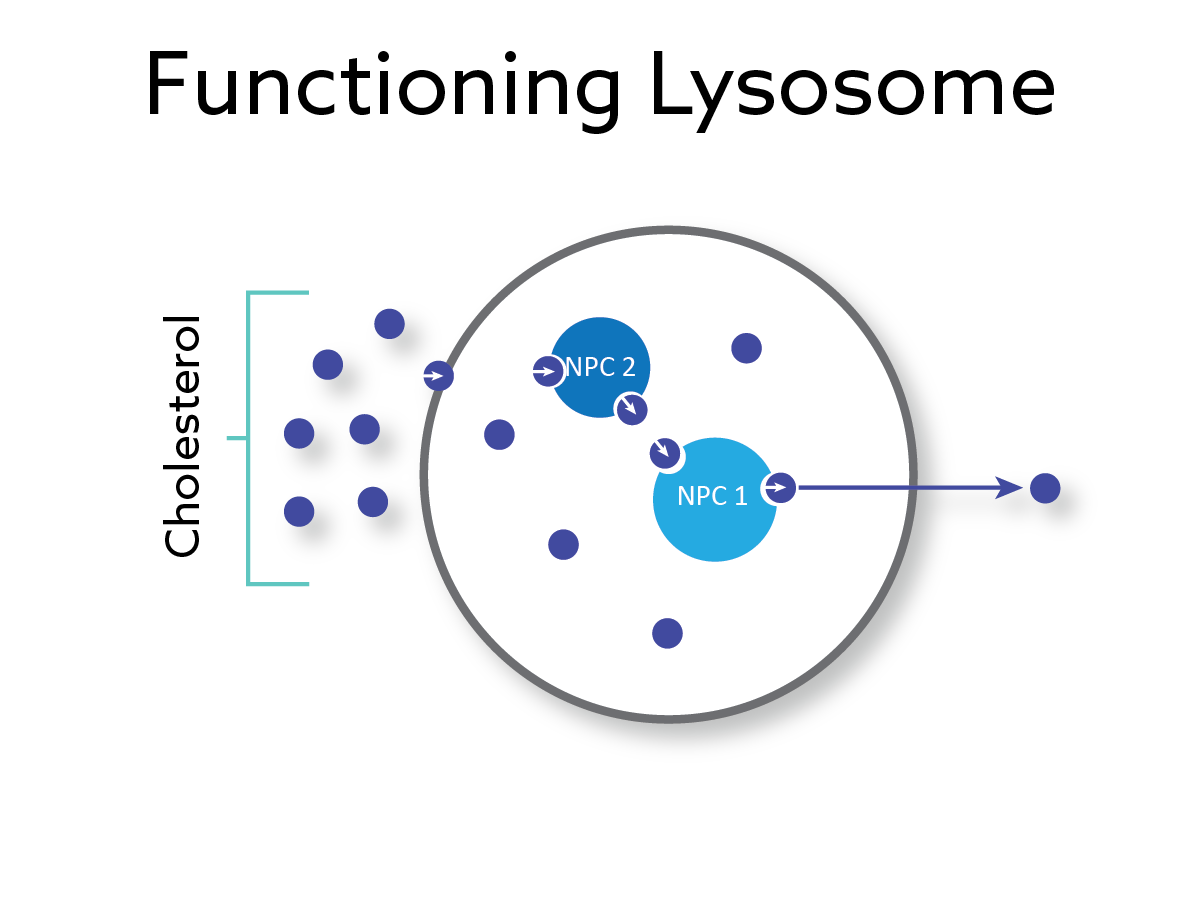
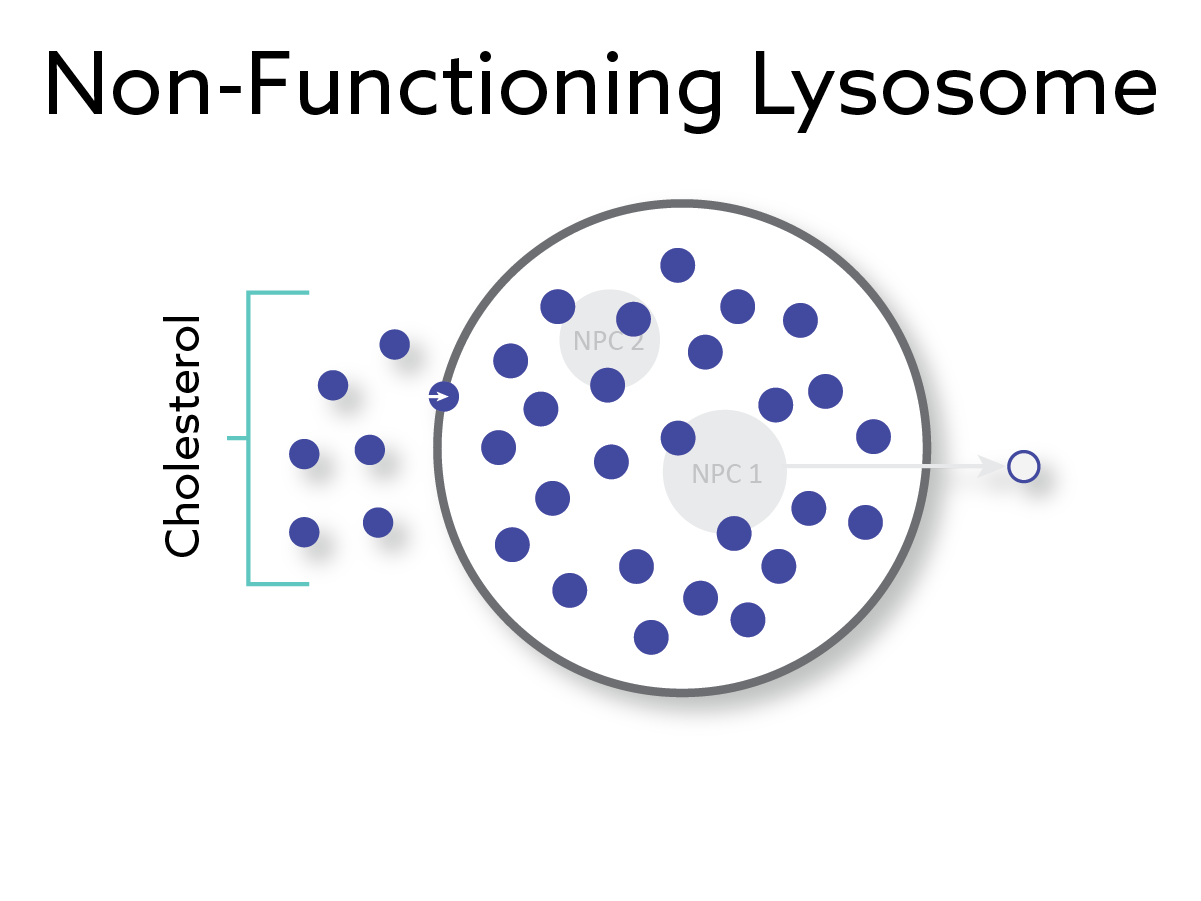
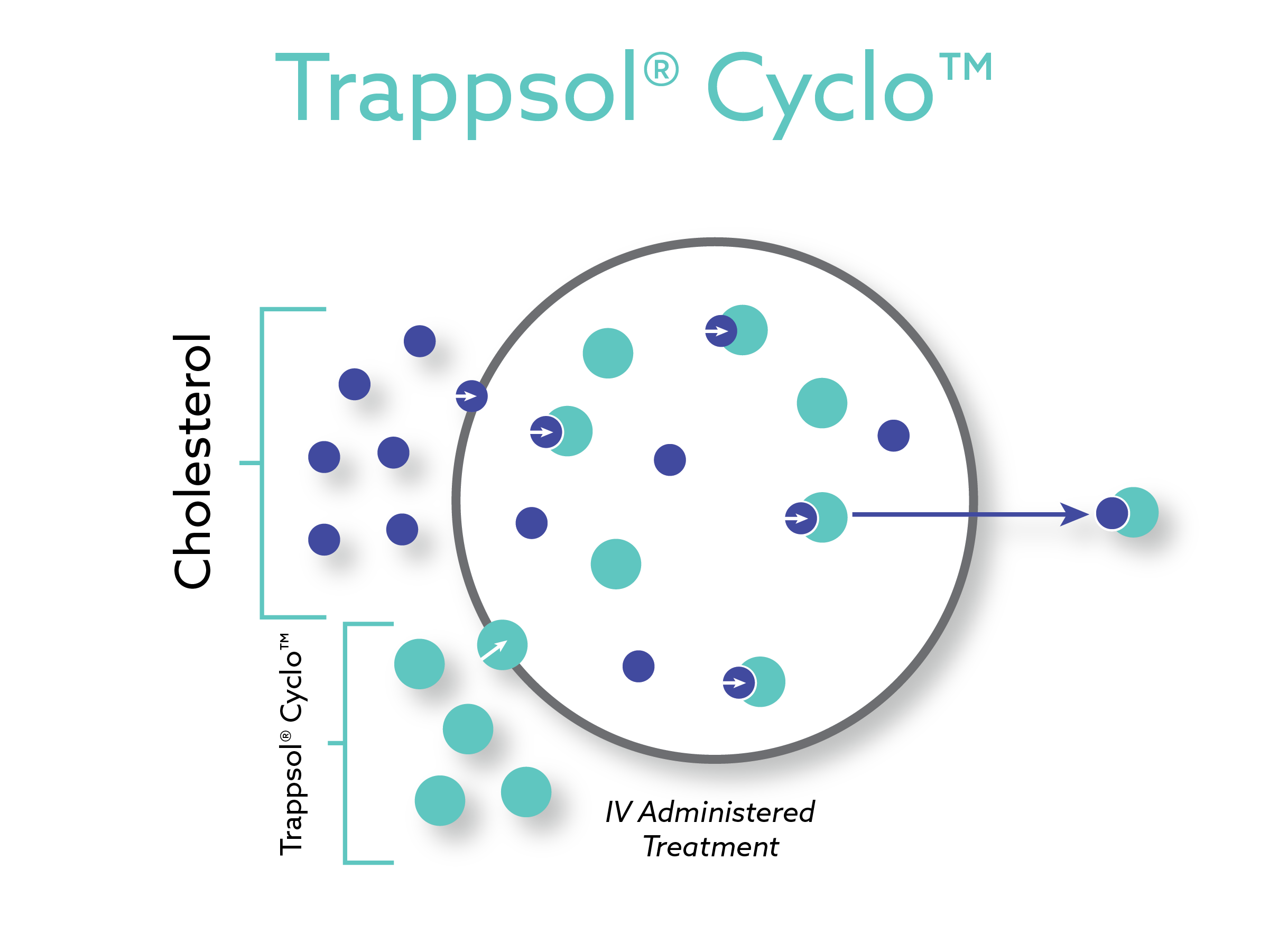
Phase 1/2 Positive Results Demonstrating Promising Safety and Efficacy
-
-
- 89% of patients met the efficacy outcome measure of improvement in at least 2 domains of the 17-domain NPC Severity Scale
- 100% of patients who completed the trial improved or remained stable per their treating physicians
- Demonstrated improvements in all dose groups
- Favorable safety profile across all dose groups
- Tau decreased: suggesting neuroprotective benefit
- Confirmed Trappsol® Cyclo™ crosses the blood brain barrier
- Improves neurological features of disease, including ataxia, swallow, walking, and quality of life
-
For Complete Study Details, Please View the Study Listing on clinicaltrials.gov: ClinicalTrials.gov Identifier: NCT02912793
Summary of Phase 1 and Open 102 Extension Study
-
-
- Favorable safety and tolerability profile
- Removed trapped cholesterol from cells and impacts cholesterol homeostasis
- Drug Present in Cerebrospinal Fluid (CSF)
- Decrease in Tau; suggesting neuroprotective benefit
- Increase in 24S biomarker; demonstrates removal of excess cholesterol from the brain
- Neurological improvements, higher energy, and greater focus exhibited by the patient
-
Virtual Investor: Niemann-Pick Disease Type C Spotlight
Trappsol® Cyclo™ is not currently approved for any indication.
Third-party websites are provided for convenience only. Cyclo Therapeutics, LLC does not approve of, or endorse any of the content. Cyclo Therapeutics, LLC does not maintain, control or monitor the content of third-party websites in any way.
