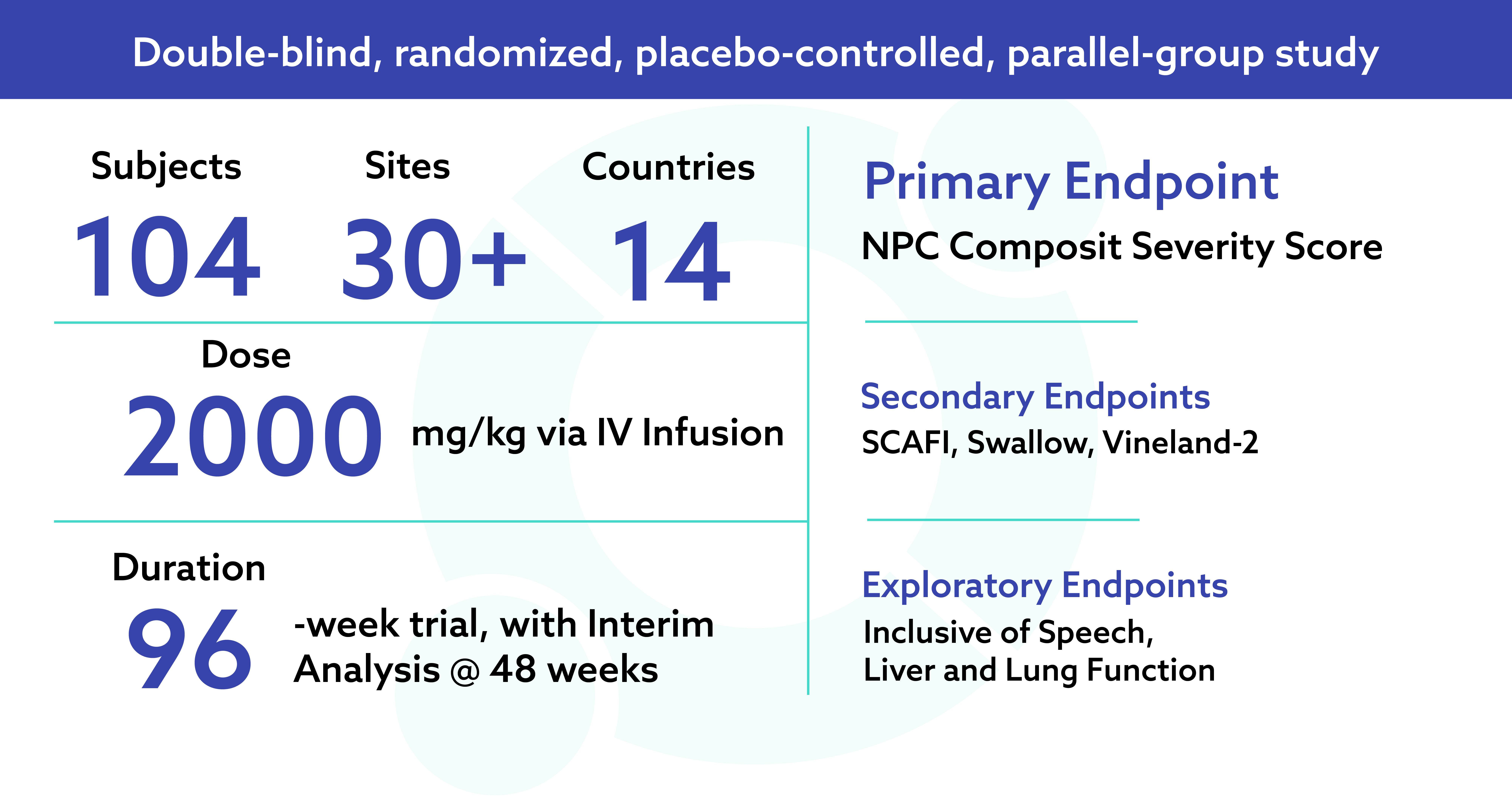
Phase 3 Study in Niemann-Pick Disease Type C

Phase 3 Clinical Trial Evaluating Trappsol® Cyclo™ in Pediatric and Adult Patients with Niemann-Pick Disease Type C1
Trial fully enrolled with 96 in core study; 10 in Substudy for ages 0-3 years.
For Complete Study Details, Please View the Study Listing on clinicaltrials.gov: ClinicalTrials.gov Identifier: NCT04860960
Patients, family members, caregivers or patient advocates looking for additional information may email us at patients@cyclotherapeutics.com.
For any other inquiries, please see
our Contact Us page.
If you or a family member are interested in participating in our ongoing study, please visit or speak with your treating physician. You can also contact us at:
clinicaltrials@cyclotherapeutics.com.
Trappsol® Cyclo™ is not currently approved for any indication.
Third-party websites are provided for convenience only. Cyclo Therapeutics, LLC does not approve of, or endorse any of the content. Cyclo Therapeutics, LLC does not maintain, control or monitor the content of third-party websites in any way.
